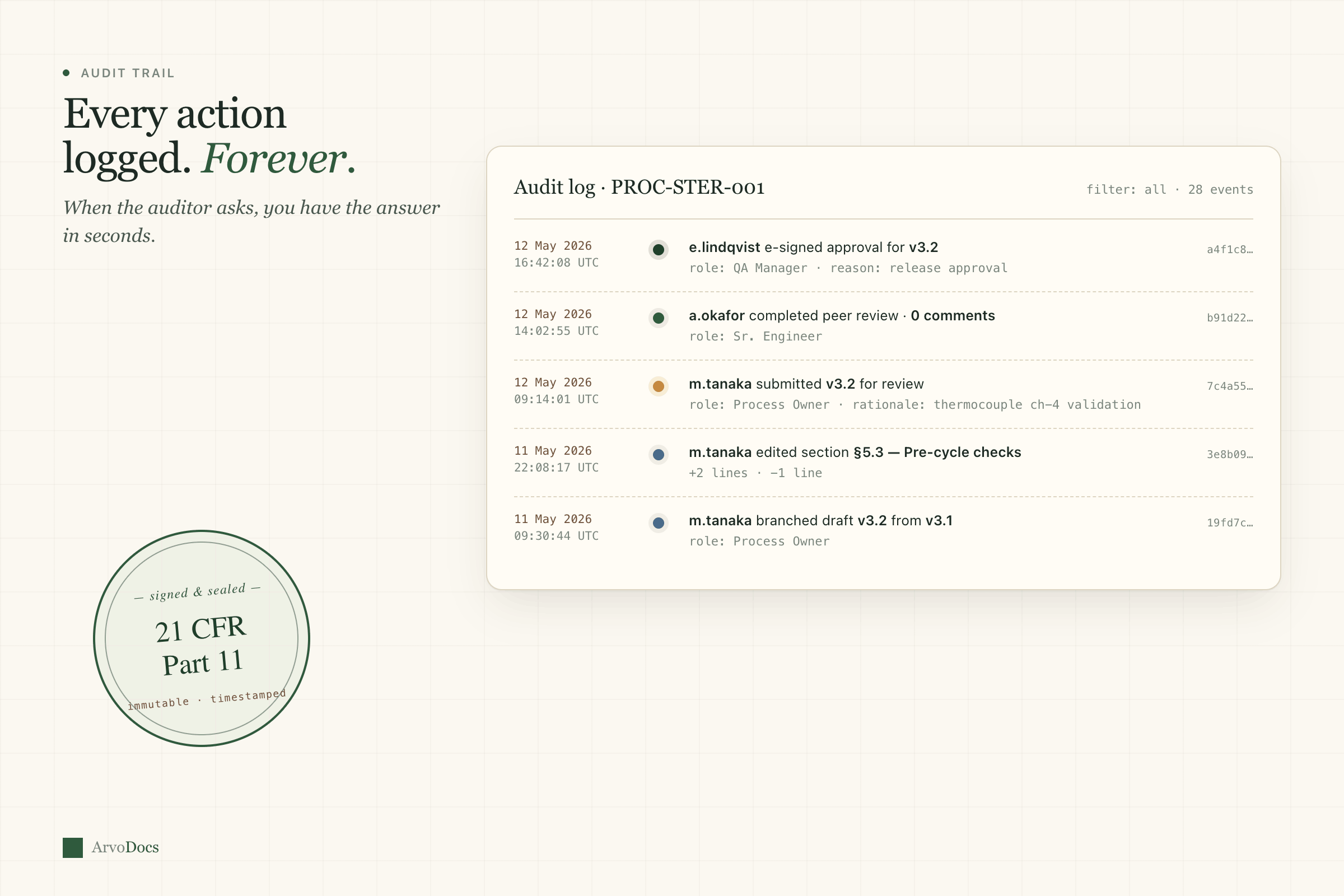
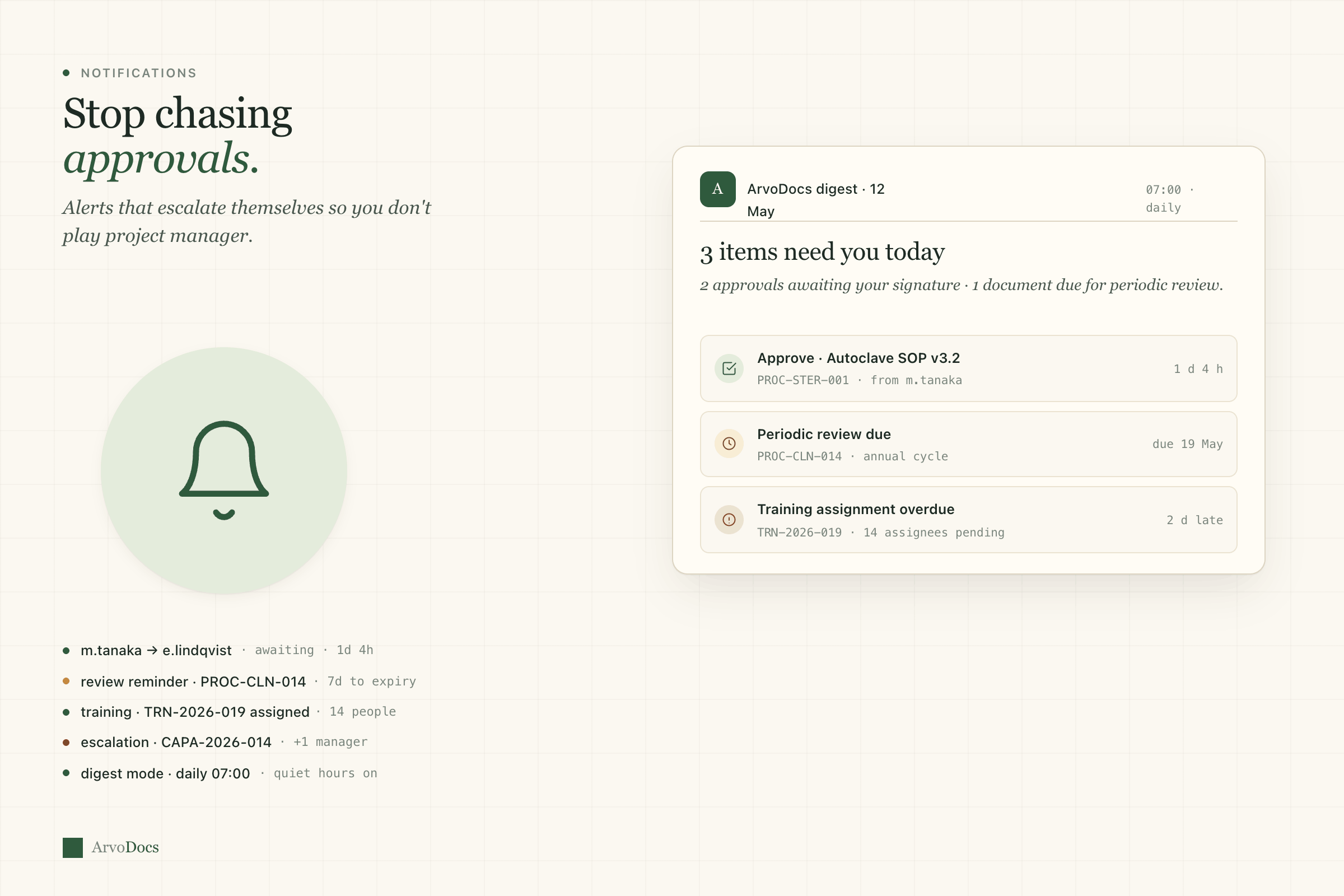
Document control
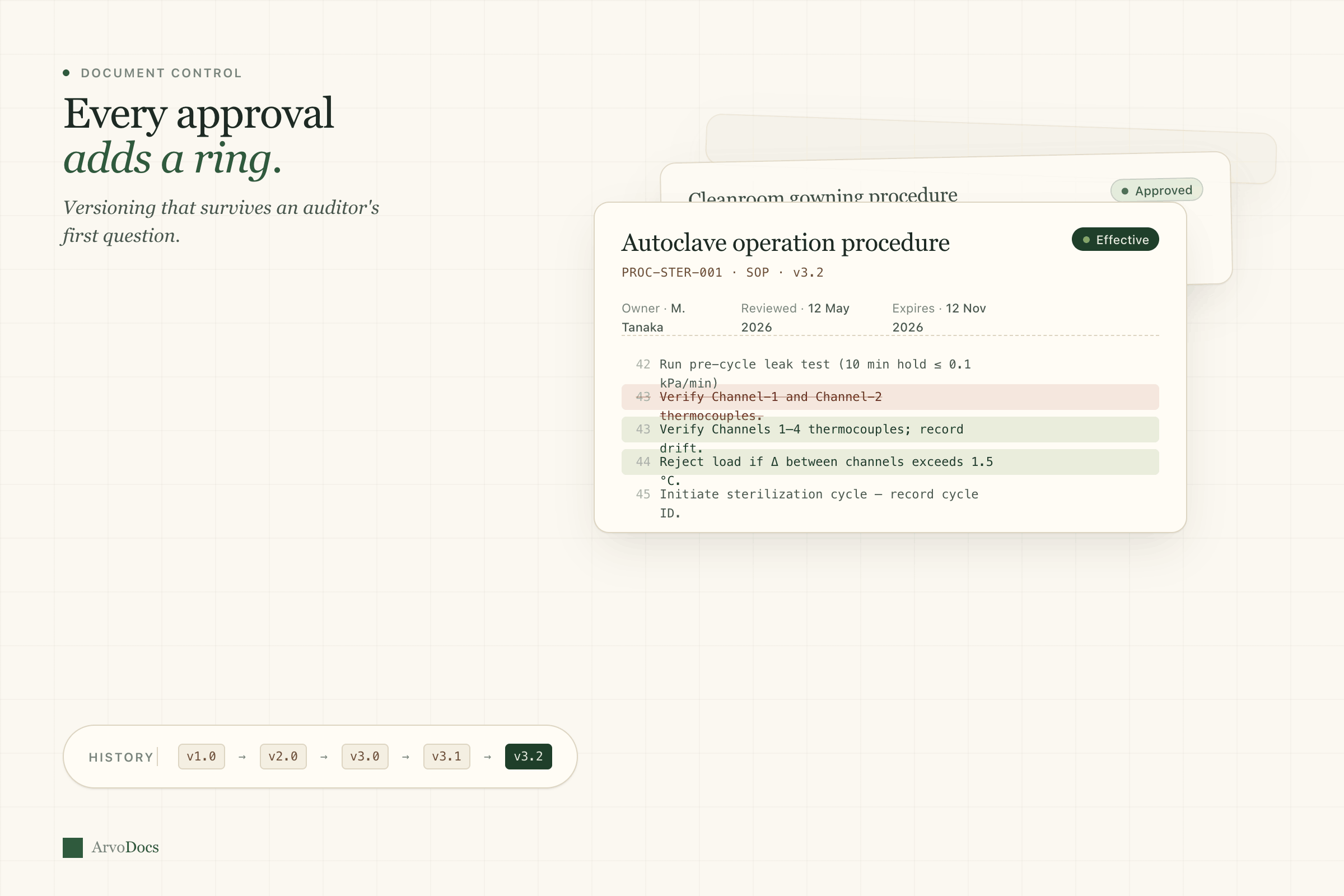
The backbone of any QMS. Version control that actually works, with approval workflows that don't make you want to throw your laptop.
- Full version history with diff view between any two revisions
- Configurable approval workflows — sequential or parallel
- Periodic review scheduling with automated reminders
- Document numbering with custom schemas
- Watermarked PDF generation for controlled copies
- Bulk import from existing file systems